A) 29
B) 30
C) 60
D) 150
E) 342
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
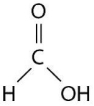 Figure 2.5
Figure 2.5 shows a representation of formic acid.A formic acid molecule
Figure 2.5
Figure 2.5 shows a representation of formic acid.A formic acid molecule
A) will dissociate in water,thus increasing the pH.
B) will dissociate in water,thus decreasing the pH.
C) contains primarily nonpolar covalent bonds.
D) has a tetrahedral shape.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If a salamander clings to surfaces through hydrogen bonds,it would have the most difficulty clinging to which of the following surfaces?
A) a surface coated with a thin film of water
B) a surface coated with a thin film of vinegar (acetic acid)
C) a surface coated with a thin film of vegetable oil
D) a surface coated with a thin film of ammonia (NH3)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
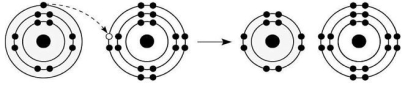 Figure 2.6
-What is the atomic number of the cation formed in the reaction illustrated in Figure 2.6?
Figure 2.6
-What is the atomic number of the cation formed in the reaction illustrated in Figure 2.6?
A) 1
B) 8
C) 10
D) 11
E) 16
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Glucose has a molecular mass of 180 g/mol.How many glucose molecules are present in 100 grams of glucose?
A) 100 × 1023
B) (6.02/180) × 1023
C) (6.02/100) × 1023
D) (100 × 6.02) × 1023
E) (100/180) × 6.02 × 1023
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Measurements show that the pH of a particular lake is 4.0.What is the hydrogen ion concentration of the lake?
A) 4.0 M
B) 10−4 M
C) 104 M
D) 10−10 M
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electron pairs are shared between the two carbon atoms in a molecule that has the formula C2H6?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One liter of a solution of pH 8 has how many more hydroxyl ions (OH-) than 1 L of a solution of pH 4?
A) 5 times more
B) 32 times more
C) 50,000 times more
D) 10,000 times more
E) 100,000 times more
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many molecules of glycerol (C3H8O3;molecular mass = 92) are present in 0.5 L of a 1 M glycerol solution?
A) 1 × 1023
B) 0) 5 × 6.02 × 1023
C) 92/2 × 6.02 × 1023
D) 0) 5 × 6.02/92 × 1023
E) 6) 02 × 1023
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The left-to-right position of an element in the first three rows of the periodic table indicates which of the following properties of the element?
A) the total number of electrons in the element
B) the total number of protons in the element
C) the total number of neutrons in the element
D) the number of electron orbitals in the element
E) the number of electrons in the valence shell
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Increased atmospheric CO2 concentrations will have what effect on seawater?
A) Seawater will become more acidic,and bicarbonate concentrations will decrease.
B) Seawater will become more alkaline,and carbonate concentrations will decrease.
C) There will be no change in the pH of seawater because carbonate will turn to bicarbonate.
D) Seawater will become more acidic,and carbonate concentrations will decrease.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If acid rain has lowered the pH of a particular lake to pH 4.0,which of the following statements about this lake is true?
A) The hydrogen ion concentration is 1 × 10-10 moles per liter of lake water.
B) The hydrogen ion concentration is 4.0 moles per liter of lake water.
C) The hydrogen ion concentration is 1 × 104 moles per liter of lake water.
D) The hydroxyl ion concentration is 1 × 10-10 moles per liter of lake water.
E) The hydroxyl ion concentration is 1 × 10-4 moles per liter of lake water.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One mole (mol) of glucose (molecular mass = 180 daltons) is
A) 180 × 1023 molecules of glucose.
B) 1 kg of glucose dissolved in 1 L of solution.
C) the largest amount of glucose that can be dissolved in 1 L of solution.
D) 180 grams of glucose.
E) 180 grams of glucose dissolved in 1 L of solution.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly describes any reaction that has reached chemical equilibrium?
A) The concentration of the reactants equals the concentration of the products.
B) The rate of the forward reaction is equal to the rate of the reverse reaction.
C) All of the reactants have been converted to the products of the reaction.
D) All of the products have been converted to the reactants of the reaction.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molar mass of glucose is 180 g/mol.Which of the following procedures should you carry out to make a 1 M solution of glucose?
A) Dissolve 1 g of glucose in 1 L of water.
B) Dissolve 180 g of glucose in 1 L of water.
C) Dissolve 180 g of glucose in 180 g of water.
D) Dissolve 180 milligrams (mg) of glucose in 1 L of water.
E) Dissolve 180 g of glucose in 0.8 L of water,and then add more water until the total volume of the solution is 1 L.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the atoms shown would be most likely to form a cation with a charge of +1?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A beaker contains 100 mL of NaOH solution at pH = 13.A technician carefully pours into the beaker 10 mL of HCl at pH = 1.Which of the following statements correctly describes the results of this mixing?
A) The concentration of Na+ ion will rise.
B) The concentration of Cl- ion will be 0.1 M.
C) The concentration of undissociated H2O molecules will remain unchanged.
D) The pH of the beaker's contents will be neutral.
E) The pH of the beaker's contents will decrease.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many glucose molecules are contained in one liter of a 0.1 M solution of glucose in water?
A) 6) 02 × 1023
B) 3) 01 × 1023
C) 6) 02 × 1024
D) 12.04 × 1023
E) 6) 02 × 1022
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why does ice float in liquid water?
A) The high surface tension of liquid water keeps the ice on top.
B) The ionic bonds between the molecules in ice prevent the ice from sinking.
C) Ice always has air bubbles that keep it afloat.
D) Hydrogen bonds stabilize and keep the water molecules of ice farther apart than the water molecules of liquid water.
E) The crystalline lattice of ice causes it to be denser than liquid water.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The attraction between the slight negative charge of one water molecule to the slight positive charge of another water molecule results in which of the following interactions?
A) a covalent bond
B) a hydrogen bond
C) an ionic bond
D) a hydrophilic bond
E) a van der Waals interaction
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 135
Related Exams